Sp3 sp3d

Available here Image Courtesy:ġ.”AE2h”By Jfmelero – Own work, (CC BY-SA 3.0) via Commons WikimediaĢ.”AE3h”By Jfmelero – Own work, (CC BY-SA 3.0) via Commons Wikimediaģ.”AE4h”By Jfmelero – Own work, (CC BY-SA 3.The content that follows is the substance of General Chemistry Lecture 35. “Hybridization.” Chemistry LibreTexts, Libretexts, 21 July 2016. “Orbital Hybridisation.” Wikipedia, Wikimedia Foundation, 13 Sept. The key difference between sp sp2 and sp3 is that the sp hybrid orbitals have 50% s orbital characteristics and the sp2 hybrid orbitals have 33% s orbital characteristics whereas the sp3 hybrid orbitals have 25 % s orbital characteristics. The simplest forms of atomic orbital hybridizations are sp, sp2 and sp3 hybridizations. Hybridization is a process in which atomic orbitals mix with each other to form new hybrid orbitals that can undergo covalent chemical bonding. The below infographic shows a detailed side by side comparison on the difference between sp sp2 and sp3 hybridization. Thus, it leaves two un-hybridized p atomic orbitals. For example, the sp hybridization involves only 1 p atomic orbitals. Moreover, each hybridization leaves different numbers of un-hybridized orbitals.
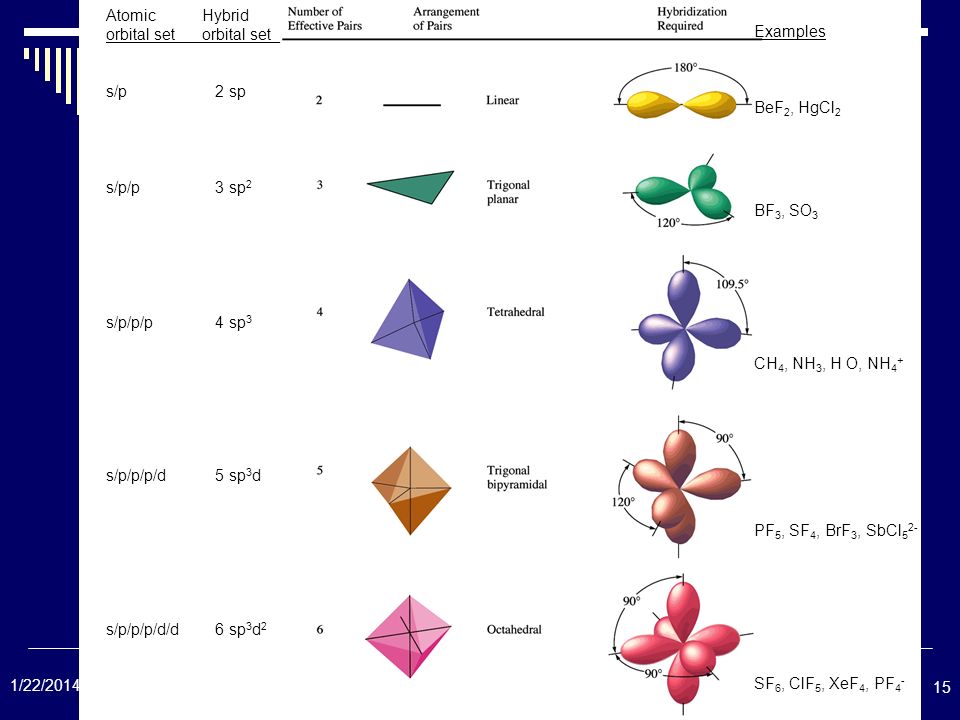
However, we can emphasize the major difference between sp sp2 and sp3 hybridization according to the s characteristic of these hybrid orbitals sp hybrid orbitals have 50% s orbital characteristics, and sp2 hybrid orbitals have 33% s orbital characteristics whereas sp3 hybrid orbitals have 25 % s orbital characteristics. Therefore they have different p orbital characteristics as well. Moreover, each of the new hybrid orbitals that form in these three forms of hybridizations has different s orbital characteristics because the s orbitals mix with different numbers of p orbitals. This is the fundamental difference between sp sp2 and sp3 hybrid orbitals. Sp hybridization is the simplest form of hybridization in which an s orbital overlaps with a p orbital to form two new sp orbitals, and an Sp2 hybridization is a form of orbital hybridization in which one s orbital overlaps with two p orbitals to form three new hybrid orbitals whereas Sp3 hybridization is a form of orbital hybridization in which one s orbital overlaps with three p orbitals. What is the Difference Between sp sp2 and sp3? These new hybrid orbitals arrange in a tetrahedral arrangement with 109.5◦ bond angle. Since the ratio between s and p orbitals is 1:3, the s characteristic of each hybrid orbital is 25% while the p orbital characteristic is 75%. Hence, this results in 4 new hybrid orbitals. Therefore, there are two un-hybridized p orbitals remaining in these atoms.įigure 03: Spatial Arrangement of sp3 Hybrid Orbitals In this hybridization, one of these three p orbitals mixes with an s orbital of the same atom. An electron shell contains three p orbitals. Sp hybridization is the simplest form of hybridization in which an s orbital overlaps with a p orbital to form two new sp orbitals. Side by Side Comparison – sp vs sp2 vs sp3 in Tabular Form Sp, sp2 and sp3 are some common hybridizations that involve in the s and p orbitals of an atom.

There are several forms of hybridizations according to the atomic orbitals that take part in the hybridization process. These orbitals can undergo hybridization in order to form new hybrid orbitals that can form covalent chemical bonds. Orbitals are hypothetical regions around a nucleus of an atom, which contain electrons of that atom. The terms sp, sp2 and sp3, refer to different hybridizations of orbitals that leads to form hybrid orbitals. The key difference between sp sp2 and sp3 is that the sp hybrid orbitals have 50% s orbital characteristics and sp2 hybrid orbitals have 33% s orbital characteristics whereas sp3 hybrid orbitals have 25 % s orbital characteristics.
